UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
Date of Report (Date of earliest event
reported):
(Exact name of registrant as specified in its charter)
| (State or other Jurisdiction of Incorporation) |
(Commission File Number) | (IRS Employer Identification No.) |
(Address of principal executive offices)(Zip Code)
Registrant’s telephone number, including area code: (
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 7.01 Regulation FD Disclosure
On January 10, 2023, AbbVie Inc. posted an investor presentation to its website at: https://investors.abbvie.com/presentations. A copy is attached as Exhibit 99.1 to this Current Report on Form 8-K.
As provided in General Instruction B.2 of Form 8-K, the information in this Item 7.01 and Exhibit 99.1 incorporated herein shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, nor shall they be deemed to be incorporated by reference in any filing under the Securities Act of 1933, as amended, except as shall be expressly set forth by specific reference in such a filing. Additionally, the submission of the information set forth in this Item 7.01 is not deemed an admission as to the materiality of any information in this Current Report on Form 8-K that is required to be disclosed solely by Regulation FD.
Item 9.01 Financial Statements and Exhibits
(d) Exhibits
| Exhibit No. | Exhibit | |
| 99.1 | Investor Presentation dated January 10, 2023. | |
| 104 | The cover page from this Current Report on Form 8-K formatted in Inline XBRL (included as Exhibit 101). |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| ABBVIE INC. | |||
| Date: January 10, 2023 | By: | /s/ Scott T. Reents | |
| Scott T. Reents | |||
| Executive Vice President, Chief Financial Officer | |||
| J.P. Morgan Healthcare Conference | January 10, 2023 1 J.P. Morgan Healthcare Conference Rick Gonzalez Chairman and Chief Executive Officer January 10, 2023 |
| J.P. Morgan Healthcare Conference | January 10, 2023 2 Forward - Looking Statements and Non - GAAP Financial Information Some statements in this presentation are, or may be considered, forward - looking statements for purposes of the Private Securitie s Litigation Reform Act of 1995. The words “believe,” “expect,” “anticipate,” “project” and similar expressions and uses of future or cond iti onal verbs, generally identify forward - looking statements. AbbVie cautions that these forward - looking statements are subject to risks and un certainties that may cause actual results to differ materially from those expressed or implied in the forward - looking statements. Such risks and uncertainties include, but are not limited to, challenges to intellectual property, competition from other products, difficul tie s inherent in the research and development process, adverse litigation or government action, and changes to laws and regulations applicable to our industry. Additional information about the economic, competitive, governmental, technological and other factors that may affect AbbVie’ s o perations is set forth in Item 1A, “Risk Factors,” of AbbVie's 2021 Annual Report on Form 10 - K, which has been filed with the Securities and Exchange Commission, as updated by its Quarterly Reports on Form 10 - Q and in other documents that AbbVie subsequently files with the Secu rities and Exchange Commission that update, supplement or supersede such information. AbbVie undertakes no obligation, and specifica lly declines, to release publicly any revisions to forward - looking statements as a result of subsequent events or developments, exce pt as required by law. This presentation contains GAAP and certain non - GAAP financial measures. Non - GAAP financial measures are adjusted for certain no n - cash items and for factors that are unusual or unpredictable, and exclude those costs, expenses and other specified items pre sen ted in AbbVie’s reconciliation tables. AbbVie’s management believes non - GAAP financial measures provide useful information to investors regarding AbbVie’s results of operations and assist management, analysts and investors in evaluating the performance of the b usi ness. Non - GAAP financial measures should be considered in addition to, and not as a substitute for, measures of financial performance p rep ared in accordance with GAAP. Reconciliations of these non - GAAP financial measures to the most comparable GAAP measures are available in the appendix to this presentation and on the company’s website at www.abbvieinvestor.com. Today’s discussions and presentation are intended for the investor community only; materials are not intended to promote the pro ducts referenced herein or otherwise influence healthcare prescribing decisions. |
| J.P. Morgan Healthcare Conference | January 10, 2023 3 Not for promotional use AbbVie is a Diversified Biopharmaceutical Company with Multiple Sources of Growth Immunology Oncology Neuroscience Aesthetics Best - in - Class medicines position AbbVie for sustained leadership; Significant growth potential for Skyrizi and Rinvoq Large, established position in blood cancer; Building a broad pipeline across a range of cancers for long - term growth Positioned for significant growth with attractive commercial opportunities across Migraine, Psychiatry Neuro - Degeneration Leadership positions across core areas; New products, increasing penetration, and global expansion drive significant long - term growth Eye Care Large franchise with investment opportunities to sustain leadership; Drive growth through internal and external innovation Clear Path to Long - Term Growth Following U.S. Humira LOE; Expect Return to Strong Top - Line Growth in 2025, with High - Single Digit CAGR Through Remainder of Decade |
| J.P. Morgan Healthcare Conference | January 10, 2023 4 Not for promotional use A Unique Investment Opportunity With Strong Cash Flow to Support Capital Allocation Priorities Compelling capital allocation philosophy, balanced between supporting growth and returning capital to shareholders Continued R&D investment in promising, innovative science across therapeutic categories, as well as capacity to pursue additional pipeline assets to augment the internal pipeline Committed to a robust and growing dividend; Increased quarterly dividend by 270% since inception Continued debt repayment to extinguish incremental debt associated with Allergan transaction by end of 2023; Expect to pay another $4B in maturities in 2023, bringing cumulative debt pay down to ~$34B |
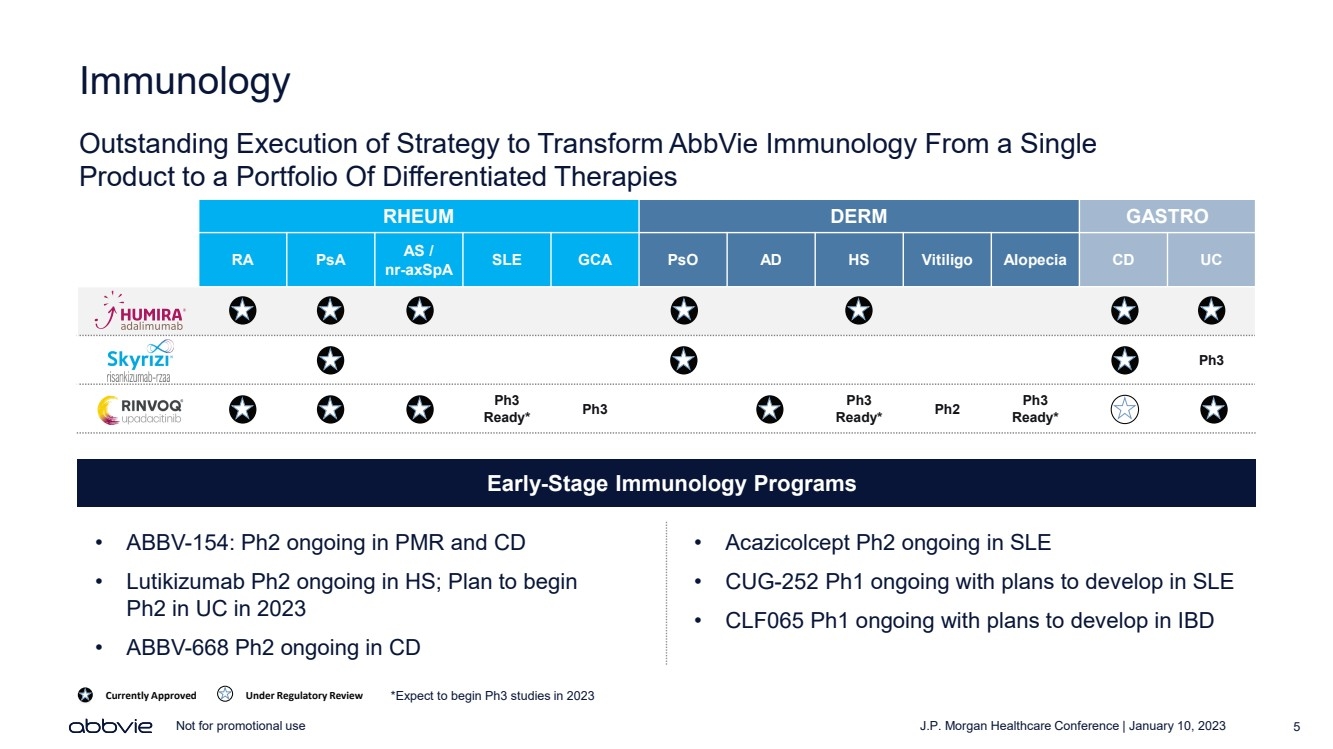
| J.P. Morgan Healthcare Conference | January 10, 2023 5 Not for promotional use Immunology Outstanding Execution of Strategy to Transform AbbVie Immunology From a Single Product to a Portfolio Of Differentiated Therapies RHEUM DERM GASTRO RA PsA AS / nr - axSpA SLE GCA PsO AD HS Vitiligo Alopecia CD UC Ph3 Ph3 Ready* Ph3 Ph3 Ready* Ph2 Ph3 Ready* Currently Approved Under Regulatory Review *Expect to begin Ph3 studies in 2023 Early - Stage Immunology Programs • ABBV - 154: Ph2 ongoing in PMR and CD • Lutikizumab Ph2 ongoing in HS; Plan to begin Ph2 in UC in 2023 • ABBV - 668 Ph2 ongoing in CD • Acazicolcept Ph2 ongoing in SLE • CUG - 252 Ph1 ongoing with plans to develop in SLE • CLF065 Ph1 ongoing with plans to develop in IBD |
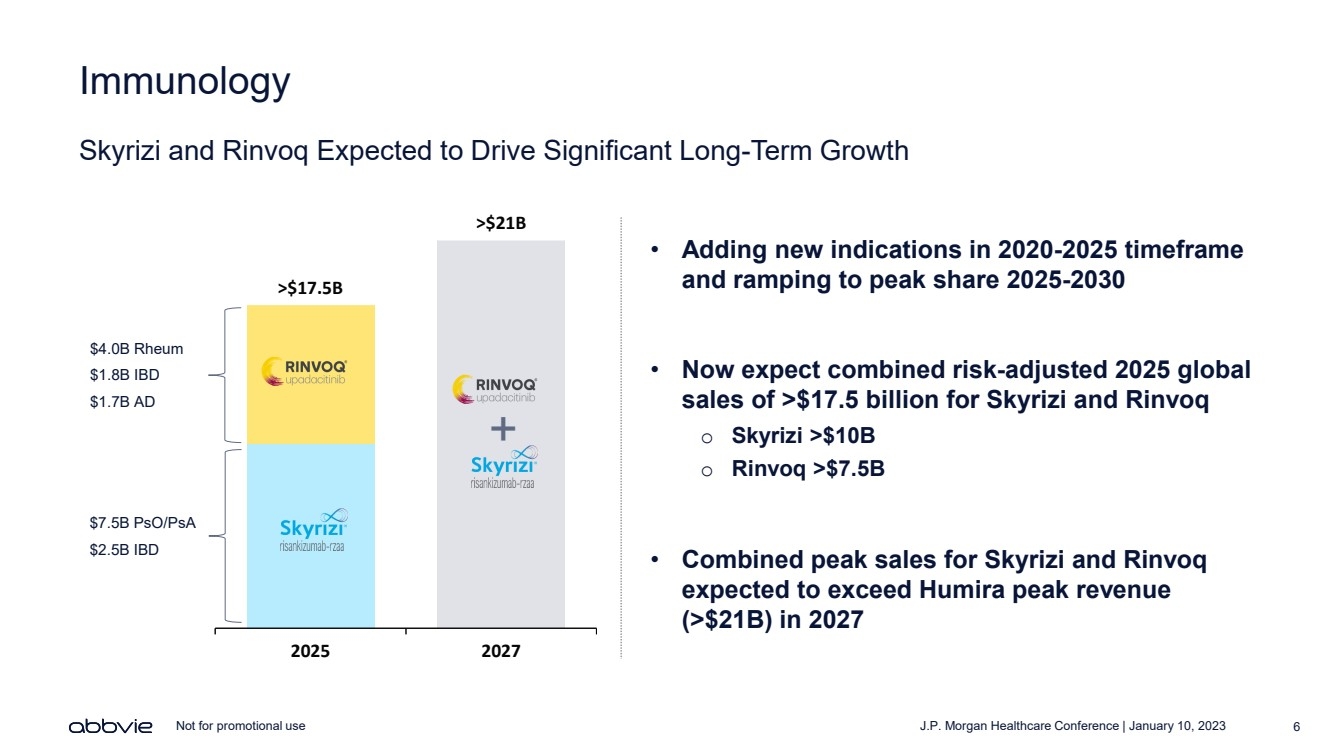
| J.P. Morgan Healthcare Conference | January 10, 2023 6 Not for promotional use Immunology Skyrizi and Rinvoq Expected to Drive Significant Long - Term Growth >$ 17.5 B >$ 21 B 2025 2027 • Adding new indications in 2020 - 2025 timeframe and ramping to peak share 2025 - 2030 • Now expect combined risk - adjusted 2025 global sales of >$17.5 billion for Skyrizi and Rinvoq o Skyrizi >$10B o Rinvoq >$7.5B • Combined peak sales for Skyrizi and Rinvoq expected to exceed Humira peak revenue (>$21B) in 2027 $4.0B Rheum $1.8B IBD $1.7B AD $7.5B PsO/PsA $2.5B IBD + |
| J.P. Morgan Healthcare Conference | January 10, 2023 7 Not for promotional use Oncology On - Market Oncology Portfolio Key Focus and Priorities: Maximize Imbruvica value in the face of increasing market and competitive challenges • E xpected to remain a key asset generating significant cash flow Strengthen Venclexta’s position in CLL / AML and position for long - term growth by broadening indications • Expect continued share gains in CLL and AML(unfit); Indication expansion in MM, AML(fit), MDS represent meaningful growth drivers Expect global oncology revenue to decline to ~$5.7B in 2023 and remain relatively flat through 2024/2025, followed by a return to growth in 2026 as new oncology products and indications ramp Established Leader in Hematologic Oncology |
| J.P. Morgan Healthcare Conference | January 10, 2023 8 Not for promotional use Oncology Oncology Pipeline Key Focus and Priorities: Successfully develop and launch new assets to support return to growth for Oncology portfolio • Epcoritamab - potential best - in - class CD3 - CD20 bispecific across B cell malignancies; anticipate initial approval in 3L+ DLBCL in 2023 * • Navitoclax - potential to improve symptoms, reverse fibrosis and modify the course of myelofibrosis; anticipate approval in 2024 • Teliso - V - potential to become an important new treatment option in non - small cell lung cancer; anticipate approval in 2L+ NSCLC in 2024 * Advance early - stage pipeline of novel heme and solid tumor assets to support sustainable, long - term growth • Focused on bi - specific, ADC platform and next - generation immuno - oncology approaches Strong Commitment to Advance Pipeline of Innovative Cancer Therapies *Anticipate Accelerated Approval based on registration - enabling Ph2 trials |
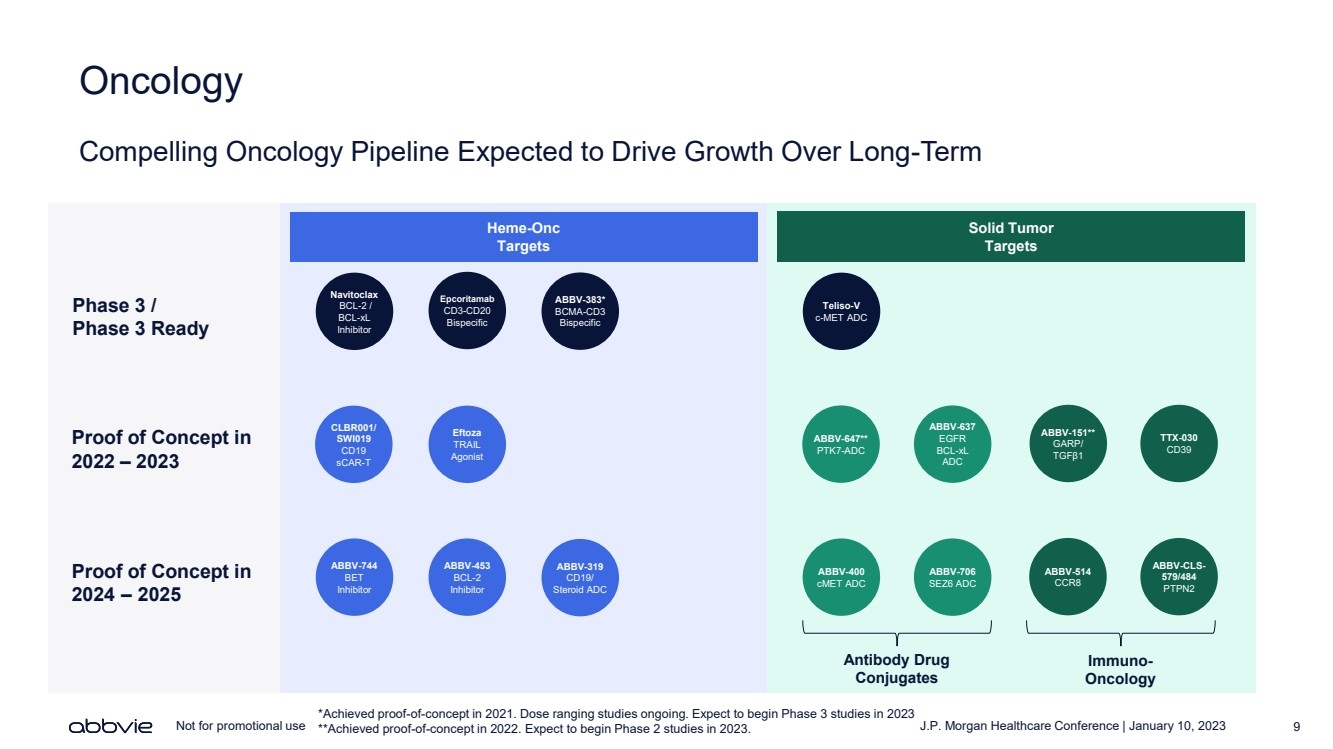
| J.P. Morgan Healthcare Conference | January 10, 2023 9 Not for promotional use Oncology Compelling Oncology Pipeline Expected to Drive Growth Over Long - Term Heme - O nc Targets Navitoclax BCL - 2 / BCL - xL Inhibitor Epcoritamab CD3 - CD20 Bispecific ABBV - 383* BCMA - CD3 Bispecific ABBV - 744 BET Inhibitor ABBV - 453 BCL - 2 Inhibitor Proof of Concept in 2022 – 2023 Proof of Concept in 202 4 – 2025 Phase 3 / Phase 3 Ready Solid Tumor Targets ABBV - 151** GARP/ TGF β 1 TTX - 030 CD39 ABBV - 514 CCR8 ABBV - CLS - 579/484 PTPN2 ABBV - 647** PTK7 - ADC ABBV - 400 cMET ADC Teliso - V c - MET ADC CLBR001/ SWI019 CD19 sCAR - T ABBV - 319 CD19/ Steroid ADC ABBV - 637 EGFR BCL - xL ADC ABBV - 706 SEZ6 ADC *Achieved proof - of - concept in 2021. Dose ranging studies ongoing. Expect to begin Phase 3 studies in 2023 **Achieved proof - of - concept in 2022. Expect to begin Phase 2 studies in 2023. Antibody Drug Conjugates Immuno - Oncology Eftoza TRAIL Agonist |
| J.P. Morgan Healthcare Conference | January 10, 2023 10 Not for promotional use Neuroscience Leading Portfolio with Options for Every Patient Across the Migraine Spectrum Acute Migraine Treatment • Leading treatment for migraine attacks providing rapid and sustained pain relief, with convenient dosing • Expect >$1B in peak sales Migraine Prevention • Approved for episodic migraine; chronic migraine under regulatory review • Expect peak sales of >$1B • A unique foundational treatment for prevention of chronic migraine • Development ongoing for potential expansion into episodic migraine |
| J.P. Morgan Healthcare Conference | January 10, 2023 11 Not for promotional use Neuroscience Developing Innovative Therapies for Mood, Thought and Anxiety Disorders • Versatile atypical antipsychotic that provides strong efficacy across multiple symptoms, with minimal impact on weight, lipids and fasting blood glucose • Recently approved as an adjunctive treatment for major depressive disorder, offering an optimal combination of powerful efficacy and trusted tolerability • Now expect Vraylar peak revenue of approaching $5B Pipeline Focused on Novel Dopamine Receptor Modulators for Neuropsychiatric Conditions and SV2A Positive Modulators to Treat Cognitive Impairment in a Range of Neuropsychiatric and Neurodegenerative Disorders $ 17 B $ 32 B 2022 2027 Global Psychiatry Market * *Based on Evaluate Pharma estimated sales |
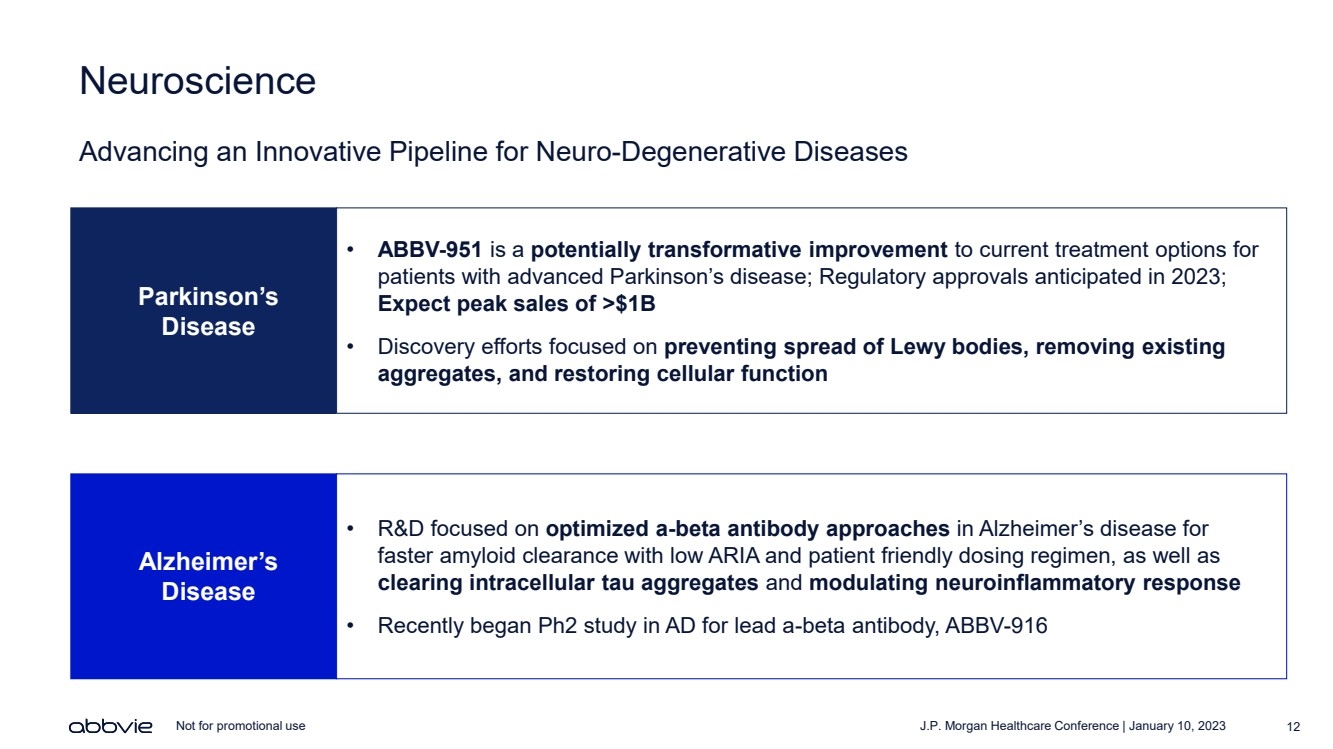
| J.P. Morgan Healthcare Conference | January 10, 2023 12 Not for promotional use Neuroscience Advancing an Innovative Pipeline for Neuro - Degenerative Diseases • ABBV - 951 is a potentially transformative improvement to current treatment options for patients with advanced Parkinson’s disease; Regulatory approvals anticipated in 2023; Expect peak sales of >$1B • Discovery efforts focused on preventing spread of Lewy bodies, removing existing aggregates, and restoring cellular function Parkinson’s Disease • R&D focused on optimized a - beta antibody approaches in Alzheimer’s disease for faster amyloid clearance with low ARIA and patient friendly dosing regimen, as well as clearing intracellular tau aggregates and modulating neuroinflammatory response • Recently began Ph2 study in AD for lead a - beta antibody, ABBV - 916 Alzheimer’s Disease |
| J.P. Morgan Healthcare Conference | January 10, 2023 13 Not for promotional use Aesthetics Market Leader in Global Aesthetics with Significant Growth Opportunities from New Product Introduction, Increasing Market Penetration and Global Expansion $14B Global Market with Low Consumer Penetration and Significant Growth Potential Market Leading Positions in Key Aesthetics Categories Strategic Investments Drive Growth • Sustained investment in consumer acquisition and retention • Enhanced digital products & services through Allē loyalty program • Best - in - class injector training program to grow base of skilled providers • International expansion to develop high growth markets including China, Japan and Latin America Continued Innovation through R&D Programs and Business Development • Innovative Toxin Pipeline: Short - and long - acting toxins • Dermal Filler Expansion: Bio - stimulatory and regenerative fillers • Body Contouring: Soliton for cellulite and CoolSculpting enhancements Expect Global Aesthetics sales of >$9B in 2029 Source: Medical Insight, Inc. Global Aesthetic Market Study November 2021 and AbbVie data. |
| J.P. Morgan Healthcare Conference | January 10, 2023 14 Not for promotional use Eye Care Eye Care Portfolio Key Focus and Priorities: Strong Foundation with Broad Portfolio Across Retina, Glaucoma, Refractive, Dry Eye and Consumer Eye Care Generates Significant Cash Flow for Investment • Enhance standard - of - care in chronic retinal conditions with RGX - 314, a potential one - time gene therapy • Establish new treatments in geographic atrophy and explore vision restoration RETINA • Optimize drop - free interventions ( Durysta , Xen, Prostamide 408 SR) • Explore neuroprotective MOAs to preserve and regenerate the optic nerve GLAUCOMA • Improve standard of care through pursuit of differentiated MOAs (AGN - 242428 ROR g ) and novel sustained release platforms DRY EYE CONSUMER REFRACTIVE • Explore novel topical drops to provide enhanced benefits for presbyopia • Pursue next - generation corneal regenerative therapies • Innovate Artificial Tears with novel ingredients and new presentations • Expand into adjacent areas (e.g., allergy, ocular health) |
| J.P. Morgan Healthcare Conference | January 10, 2023 15 Not for promotional use Innovation - Driven R&D Organization Proven Track Record of Developing New Medicines That Consistently Elevate the Standard of Care 240+ Clinical Trials 5 Core Therapeutic Areas 13K+ R&D Employees 80+ Clinical Programs ~50 New Molecular Entities 75+ Conditions Treated 25 Major Product or Indication Approvals* 110+ New Study Starts Planned for 2023 *Reflects approvals since 2013 |
| J.P. Morgan Healthcare Conference | January 10, 2023 16 Not for promotional use AbbVie Pipeline As of January 10, 2023 Excludes devices in development *Partnered assets; See appendix for partnership summary ■ CUG - 252* (IL - 2 Mutein) SLE ■ CLF065* (GLP - 2 agonist) IBD ■ ABBV - 151 (GARP+TGF b 1) Solid Tumors ■ ABBV - 400 (cMet ADC) NSCLC ■ ABBV - 647* (PTK7 ADC) NSCLC ■ ABBV - 637 (EGFR BCL - xL ADC) NSCLC ■ ABBV - 706 (SEZ6 ADC) SCLC ■ ABBV - 181 (PD - 1) Solid Tumors ■ ABBV - 927 (CD40) Solid Tumors ■ JAB - 3312* (SHP2) Solid Tumors ■ TTX - 030* (CD39) Solid Tumors ■ ABBV - CLS - 579* (PTPN2) Solid Tumors ■ ABBV - CLS - 484* (PTPN2) Solid Tumors ■ ABBV - 514 (CCR8) Solid Tumors ■ Eftoza (TRAIL) Heme Tumors ■ ABBV - 319 (CD19/Steroid ADC) Heme Tumors ■ ABBV - 525 (MALT1) Heme Tumors ■ CLBR001/SWI019* ( sCAR - T) Heme Tumors ■ ABBV - 383 (CD3 - BCMA) MM ■ HPN - 217* (CD3 - BCMA) MM ■ ABBV - 453 (BCL - 2) R/R MM t(11;14) ■ ABBV - 744 (BET) MF ■ ABBV - 552 (SV2A) Alzheimer’s Disease ■ ABBV - CLS - 7262* (elF2B) Multiple Neuro ■ AGN - 241622 (Alpha2) Presbyopia ■ ABBV - 1882 (anti - PD1/anti - a4b7) HIV ■ ABBV - 903 ( MPro Inhibitor) COVID ■ ABBV - 154 (TNF - Steroid ADC) PMR ■ ABBV - 154 (TNF - Steroid ADC) CD ■ Rinvoq (JAK 1) Vitiligo ■ Rinvoq (JAK 1) SLE ■ Rinvoq (JAK 1) HS ■ Acazicolcept ALPN - 101* (ICOS/CD28) SLE ■ Lutikizumab (IL - 1 a /1 b ) HS ■ ABBV - 668 (RIPK1) UC ■ ABBV - CX - 2029* (CD71) Solid/Heme Tumors ■ ABBV - 916 (a - beta) Alzheimer’s Disease ■ Botox (SNARE) Essential Tremor ■ Elezanumab ( RGMa ) Stroke ■ Elezanumab ( RGMa ) SCI ■ AL002* (TREM2) AD ■ OnabotA X (SNARE) Glabellar Lines ■ OnabotA X (SNARE) Forehead Lines ■ AGN - 242428 ( ROR g ) Dry Eye ■ ABBV - RGX - 314* (NAV AAV8 Anti - VEGF Fab) wAMD Suprachoroidal Delivery ■ ABBV - RGX - 314* (NAV AAV8 Anti - VEGF Fab) DR Suprachoroidal Delivery ■ AGN - 193408 ( Prostamide 408 SR) Glaucoma ■ CF Triple Combo (CFTR - C1/CFTR - C2/CFTR - P) ■ Armour Thyroid (T3T4) Hypothyroidism ■ AGN - 151607 (SNARE) Atrial Fibrillation ■ Botox (SNARE) IC/BPS ■ Rinvoq (JAK 1) GCA ■ Skyrizi* (IL - 23) UC ■ Imbruvica* (BTK) 1L FL ■ Imbruvica* (BTK) R/R MCL ■ Imbruvica* (BTK) + Venclexta* (BCL - 2) R/R MCL ■ Venclexta* (BCL - 2) AML Maintenance ■ Venclexta* (BCL - 2) R/R MM t(11;14) ■ Venclexta* (BCL - 2) High Risk MDS ■ Navitoclax (BCL - 2/BCL - xL ) Myelofibrosis ■ Epcoritamab* (CD3 - CD20) 1L DLCBL ■ Epcoritamab* (CD3 - CD20) R/R DLBCL ■ Epcoritamab* (CD3 - CD20) R/R FL ■ Teliso - V* (cMet ADC) NSCLC ■ Botox (SNARE) Episodic Migraine ■ BoNT/E (SNARE) Glabellar Lines ■ Botox (SNARE) Masseter Prominence ■ Botox (SNARE) Platysma Prominence ■ ABBV - RGX - 314* (NAV AAV8 Anti - VEGF Fab) wAMD Subretinal Delivery ■ Aztreonam/Avibactam* (PBP3) Infection ■ Rinvoq (JAK 1) CD ■ Epcoritamab* (CD3 - CD20): R/R DLBCL ■ ABBV - 951 (dopamine receptor) PD ■ Qulipta (CGRP) Chronic Migraine Prevention Phase 1 Phase 2 Registrational / Phase 3 Submitted ■ Immunology ■ Oncology ■ Neuroscience ■ Aesthetics ■ Eye Care ■ Targeted Investment |
| J.P. Morgan Healthcare Conference | January 10, 2023 17 Not for promotional use Anticipated Key Pipeline Events 2023 2024 Regulatory Approvals Rinvoq CD Epcoritamab 3L R/R DLBCL (AA) ABBV - 951 Advanced PD Qulipta Chronic Migraine Prevention Skyrizi UC Venclexta 3L+ MM w/ t(11;14) Venclexta High Risk MDS Navitoclax 1L MF Teliso V 2L+ NSCLC (AA) Botox Platysma Botox Masseter Regulatory Submissions Skyrizi UC Venclexta 3L+ MM w/ t(11;14) Navitoclax 1L MF Botox Platysma Botox Masseter Rinvoq GCA Teliso - V NSCLC Venclexta High Risk MDS Navitoclax r/r MF Botox Episodic Migraine BoNT/E (Short - Acting Toxin) Glabellar Lines Ph3/Registrational Data Readouts Skyrizi UC Induction/Maintenance Venclexta Ph3 3L+ MM t(11;14) (CANOVA) Navitoclax Ph3 1L MF (Transform - 1) Epcoritamab Ph3 3L DLBCL Teliso - V Ph2 2L+ NSCLC Botox Ph3 Masseter Botox Ph3 Platysma BoNT/E (Short - Acting Toxin) Ph3 Glabellar Lines Rinvoq Ph3 GCA Venclexta Ph3 High Risk MDS Navitoclax Ph3 r/r MF (Transform - 2) Botox Ph3 Episodic Migraine ABBV - RGX - 314 Ph3 wAMD SR Early / Mid Stage Data Readouts Rinvoq Vitiligo ABBV - 154 (TNF - Steroid ADC) Ph2 PMR TTX - 030 (CD39) Ph1 Solid Tumors CLBR001/SWI019 (CD19 sCAR - T) Ph1 Heme Tumors Eftoza (Trail) Ph1 Heme Tumors ABBV - 637 (EGFR BCL - xLi ADC) Ph1 NSCLC AGN - 241622 (Alpha2) Presbyopia ABBV - 154 (TNF - Steroid ADC) Ph2 CD Induction Lutikizumab (IL - 1 a /1 b ) Ph2 HS ABBV - CLS - 579 (PTPN2) Ph1 Solid Tumors ABBV - CLS - 484 (PTPN2) Ph1 Solid Tumors ABBV - 400 (c - Met ADC) Ph1 Solid Tumors ABBV - 453 (BCL - 2) Ph1 MM t(11;14) ABBV - 744 (BET) Ph1 MF Elezanumab ( RGMa ) Ph2 Stroke ABBV - 552 (SV2A) Ph2 AD Cognition ABBV - CLS - 7262 (elF2B) Ph2 ALS As of January 10, 2023 AA = Accelerated Approval |
| J.P. Morgan Healthcare Conference | January 10, 2023 18 Not for promotional use Portfolio of leading brands in attractive and sustainable markets Pipeline of innovative, highly differentiated assets to address significant unmet needs, with potential to drive significant growth Compelling capital allocation philosophy balanced between supporting growth and returning capital to shareholders Track record of strong execution, consistently meeting or exceeding financial commitments to deliver industry leading financial performance A unique investment vehicle, offering top - tier revenue and EPS growth, significant cash flow and strong return of capital to shareholders AbbVie: A Unique Investment Opportunity Poised for Continued Strong Shareholder Returns |
|
| J.P. Morgan Healthcare Conference | January 10, 2023 20 AbbVie’s Partnered Assets • ABBV - 2029 developed by CytomX Therapeutics through clinical proof of concept and AbbVie holds option for additional development • ABBV - 647 developed in cooperation with Pfizer • ABBV - CLS - 579 / 484 / 7262 co - developed by Calico and AbbVie • Acazicolcept (ALPN - 101 ) developed by Alpine Immune Sciences through current Phase 2 study and AbbVie holds option for additional development • AL 002 developed by Alector through Phase 2 and AbbVie holds option for additional development • Aztreonam/Avibactam co - developed by Pfizer and AbbVie • CLBR 001 /SWI 019 developed by Calibr in a first - in - patient trial and AbbVie holds option for additional development • CLF 065 developed by Scripps/Calibr in a first - in - patient trial and AbbVie holds option for additional development • CUG - 252 developed by Cugene through Phase 1 b and AbbVie holds option for additional development • Epcoritamab developed in partnership with Genmab • HPN - 217 developed by Harpoon through Phase 1 / 2 and AbbVie holds option for additional development • Imbruvica jointly developed with Janssen Biotech • JAB - 3312 developed in partnership with Jacobio • RGX - 314 co - developed by REGENXBIO and AbbVie • Skyrizi developed in cooperation with Boehringer Ingelheim • Teliso - V licensed from Seagen and Pierre Fabre • TTX - 030 developed by Trishula Therapeutics through Phase 1 b and AbbVie has option to lead global development • Venclexta jointly developed with Roche • Vraylar developed in cooperation with Gedeon Richter |